- English
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
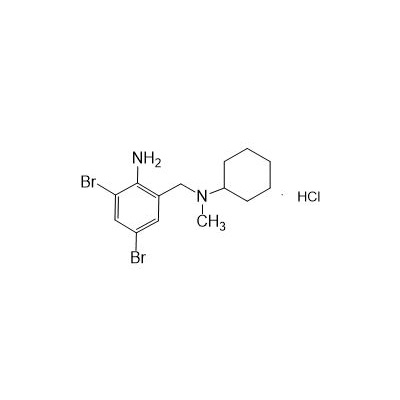
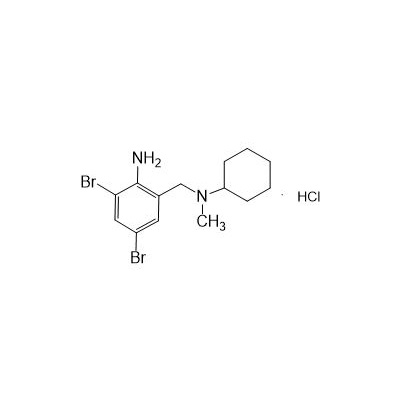
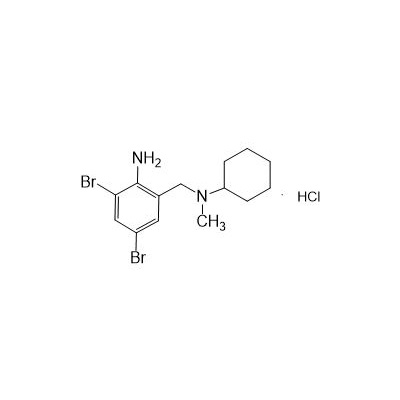
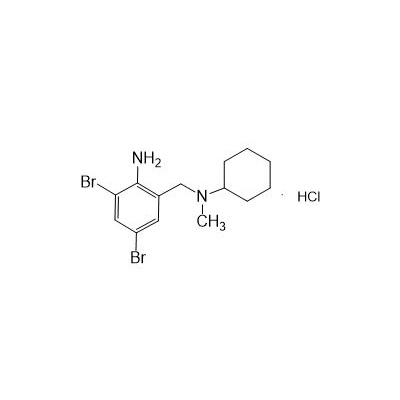
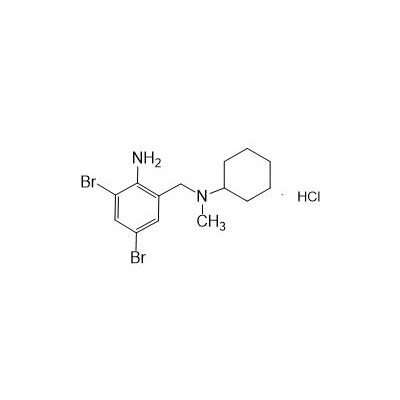
II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride
Seres Product Name: Bromhexine Hydrochloride
Seres aliases: Bromhexine hydrochloride; Hydrochloride bromhexylamine; Benzylcyclohexylamine bromide hydrochloride; II-amino-3,5-Dibromo-n-cyclohefyl, n-methylbenzylamine hydrochloride; N- (II-amino-3,5-Dibromobenzyl) -n-Methylyclohexylamine hydrochloride;
Latina Product Name: Bromhexine Hydrochloride
CAS # 611-75-6
Mitte Inquisitionem
Formula
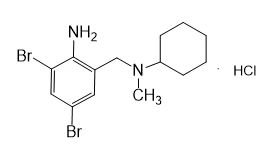
Seres Product Name: Bromhexine Hydrochloride
Seres aliases: Bromhexine hydrochloride; Hydrochloride bromhexylamine; Benzylcyclohexylamine bromide hydrochloride; II-amino-3,5-Dibromo-n-cyclohefyl, n-methylbenzylamine hydrochloride; N- (II-amino-3,5-Dibromobenzyl) -n-Methylyclohexylamine hydrochloride;
Latina Product Name: Bromhexine Hydrochloride
CAS # 611-75-6
M. Formula: C14H21BR2CLN2
M. pondus: 412.6
Et proprietatibus specie: Alba solidae
Domesticis Registration Number API: Y20170001111
Usus: usus est acuti et longos BRONCHITIS, suspiriosis, bronchiectasis, et emphysema. Praesertim idoneos qui difficultatem tussis alba glutinosus sputum et discrimine emergentia per extensive impedimentum parva bronchi sputum.
II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride est ad respiratoriorum medicamento et tussis cum pituitam pharmacum.
II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride, multifaceted compositis in modern chemiae
In realm of synthetica organicum elit et pharmaceutical investigationis, II-amino-3,5-dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride stat ex ut structuram intricate et functionally versatile compositis. Combining a brominated aromatic core cum mutatio Cyclohexylamine backbone, hoc molecule exemplificat synergy halogenation et Amine functionalization Insidijs. Et unique architectura et physicochochalicis proprietatibus habere spurata interest per diverse scientific disciplinis a medicamento inventa est materiae scientia.
I. Structural Insights et eget significant
II, amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride est characterised a II-aminobenzyl scaffold substituitur cum bromine atomos ad 3- et V, Methylamine Group. Et Hydrochloride sal enhances eius solubility et stabilitatem, faciens idoneam experimentalem et industriae applications. Key structural features includit:
Brominated aromatica anulus: et electronic, recedens bromine atomos auget electrophilicity, facilitating nucleophilic aromaticorum substitutione profectae.
Tertiariis Amine functionality: et N-Cyclohexyl-N-methyl coetus confert ad steric mole et lipophilicity, influens interactiones cum biologicis peltas.
Formation sal formation: de Amine Group improves Crystallination et Handling Properties.
Hoc compositis est M. pondus (~ 480,3 G / mol) et ratione logp (~ 3.5), suadeant libratum solubility et membrana permeability studiis et utrique synthetica manipulation et bioactive studiis.
II. Saccharum meatus et Optimization
Synthesis II-Amino-3,5-Dibromo-N-Cyclohefyl, N-Methylbenzylamine Hydrochloride typically involves sequentem Halogenation et Alkylation Steps:
II-aminobenzyl derivationes bromination: selectivam dibromination ad 3 et V, positions usura reagentia sicut n-bromosuccinimide (NBS) sub moderari conditionibus.
Amine Alkylation: reactionem de brominated medium cum n-methylyclohexylamine in conspectu de basi (E.G., K₂co₃) ad formare Tertiariis Amine next.
Formation sale curatio cum hydrochloric acidum cedere hydrochloride sal, sequitur recrymstallization pro purificatione.
Recens progressiones Leverage Proin, assisted synthesis ad redigendum reactionem temporibus et amplio cedit (usque LXXII%). Praeterea, catalytic modi usus Palladii vel aeris complexorum sunt explored ad augendae regioselectivity durante bromination.
III. Pharmacological et biologicis applications
II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride est demonstratum significant potentiale in biomedical investigationis:
Central nervosi (CNS) Targeting: Praevia Studies indicant modicum affinitate Serotonin receptores (V, ht₂c), suggerendo utilitatem in mente inordinatio inordinatio.
Antimicrobial Activity: Brothines Atoms conferre potens actio contra medicamento-repugnans Staphylococcus aureus (mic 2-4 μg / ml) per dissimulare bacterialem membranam integritatem.
Enzyme inhibition: in vitro assays revelare inhibitory effectus in dapibus kinase C (PKC), positioning eam ut candidatum ad anticancer medicamento progressionem.
Et Hydrochloride sal formam augendae Bioavailability, cum LXXXV% stabilitatem in simulatis intestinorum fluidum super XII horas, a discrimine elementum ad oralis administratione.
IV. Industrial et materialium scientia relevance
Quam Pharmacology, II-Amino-3,5-Dibromo-N-Cyclohefyl, N-Methylbenzylamine Hydrochloride Finds Utilitas in Specialized Applications:
Coordinatio Chemiae: Act ut a Ligand ad transitus metallum catalysts in crucem, coupling reactiones, leveraging eius electronic-deficiens aromaticum circulum.
Polymer additives: incorporatus in flamma-retardant Polymers debitum ad bromine scriptor radicitus-scavenging possessiones.
Analytica Signa: usus est ut referat compositis in Missam spectrometry et hplc modum progressionem.
V. Challenges et limitations
Quamvis eius promissionem, in latos adoption of II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride facies crates:
Saccharum complexionem: multi-gradus synthesis et purificatio processibus escalata productio costs.
Environmental de: Brominated componit erigere ecotoxicity exitibus, necessitating restrictius vastum procuratio protocols.
Metabolicae instabilitatem, celeri hepaticum Clearance observari in preclinical exempla requirit structuram Optimization pro diuturna efficacia.
VI. Future Spes et innovations
Ongoing Research Aims ad expand utilitatem II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride per:
Prodrug Design: Masking Amine Group ad augendae metabolicae stabilitatem et TEXTUS targeting.
Nanocarrier integration: Encapsulation in Lipid-secundum Nanoparticles ut amplio Cns partum.
Sustainable Synthesis: Developing Electrochemical Bromation modi ad redigendum halogen desertum.
II-amino-3,5-Dibromo-n-cyclohexyl, n-methylbenzylamine hydrochloride invigiendam convergence synthetica ingenii et multifunctional consilio. Et brominated aromatic systematis, copulata cum a tailored amine scaffold, offert a versatile platform pro medicamento inventa, Catalysis et materiam ipsum. Dum challenges in scalability et environmental impulsum perseverare, innovations in synthetica methodologies et applicationem-specifica modificationes tenere clavem ad reserans eius plena potential. Ut Interdisciplinary Research Advances, hoc compositis est poised ludere a Pivotal partes in addressing universa scientific et industriae challenges.